Lab 5 & 6 Hot and cold - review, algebra, and XC
Lab 5
In lab 5, the idea was to guess and test an equation for predicting the temperature of a mixture of hot and cold water.
Review
With equal amounts of hot and cold water, most of you predicted that the temperature would be half-way between the temperatures of hot and cold water.
To be more precise, let's define these quantities:
- $T_H$ is the initial temperature of the hot water (or maybe just warmer than room temperatures) before mixing. To measure this accurately, you must leave a thermometer in the hot water cup until the temperature appears to be unchanging (this usually takes under a minute after putting the thermometer in the cup).
- $T_C$ is the initial temperature of the cold water (or room temperature water).
- $T_f$ is the final temperature of the mixture once you've poured one of the cups into the other and the temperature of the thermometers has had a chance to stabilize to the water temp.
The temperature "halfway between" the two temperatures is the same as the average of the hot and cold temperatures. Most of you know how to calculate the average of two numbers like this: $$T_f=\frac{T_C+T_H}{2}.\label{equal}$$ And most of you found in the lab that mixing equal amounts of water resulted in a final temperature of the mixed waters of very nearly half-way between the hot and cold temperatures!
Mix unequal amounts of hot and cold water
Remind yourself about two mathematical facts
- You can write a fraction as the product of (1 divided by the denominator) times the numerator: $$\frac34 = 3*\frac14$$ or I could just as well write this as $\frac14*3$.
- And if you have two fractions that have the same denominator you can evaluate the sum by adding up the numbers in the numerators like this: $$ \frac 14+\frac34=\frac{1+3}{4}=\frac{4}{4} $$ and 4/4 = 1.
Look again at the expression for the average for equal amounts of water, Eq $\ref{equal}$, I could re-write that as: $$T_f=\frac{T_C+T_H}{2}=\color{blue}\frac12T_C+\frac12T_H.\label{halves}$$ Hmm, with equal amounts of cold and hot water, when I mix them together, the cold water made up half of the final mixture and the hot water made up half of the final mixture.
And the final temperature was half of the cold temperature plus half of the hot temperature.
...Maybe if I have different amounts of hot and cold, I could calculate the fraction of the final mixture that was cold and the fraction of the final mixture that was hot, and then use those fractions in place of 1/2 and 1/2 in Eq. $\ref{halves}$?!
How to calculate these fractions? Let's start with what you measured...
- $V_C$ is the volume (in milliliters) of the cold water you started with,
- $V_H$ is the volume of the hot water you started with,
- $V_f$ is the volume of the water that you end up with, once you have mixed the hot and cold water together
- ...and $V_f$ can be calculated instead of needing to measure it separately, since... $$V_f=V_C+V_H.$$
With these symbols, the fraction of water in the final mixture that started out cold would be: $$\text{fraction of cold water}=\frac{V_C}{V_f} =\frac{V_C}{V_C+V_H}.$$ So, if we had equal amounts of hot and cold water, $V_H=V_C$ and the fraction above would be $\frac{V_C}{2V_C}=\frac 12$.
and similarly the fraction of the final mixture that started out hot was: $$\text{fraction of hot water }\ =\frac{V_H}{V_f}=\frac{V_H}{V_C+V_H}.$$ and this fraction would also be $\frac 12$ for equal amounts of hot and cold water.
So, let's substitute these fractions instead of $1/2$ and $1/2$ in Eq. $\ref{halves}$:
Final temperature as a "weighted average" of the initial and final temperatures: $$ T_f=\left(\frac{V_C}{V_C+V_H}\right)T_C + \left(\frac{V_H}{V_C+V_H}\right)T_H \label{Tf} $$
Example
In the lab, you tried mixing 100 ml = $V_C$ of cold water with 50 ml = $V_H$ of hot water. The final temperature as predicted by Eq. $\ref{Tf}$ would be: $$\begineq T_f&=\left(\frac{V_C}{V_C+V_H}\right)T_C + \left(\frac{V_H}{V_C+V_H}\right)T_H\\ &=\left(\frac{100}{150}\right)T_C + \left(\frac{50}{150}\right)T_H\\ &=\left(\frac{2}{3}\right)T_C + \left(\frac{1}{3}\right)T_H\\ \endeq $$
Now you try it! (Extra Credit)
- Measure out approximately 40 ml of cold water and 100 ml of hot water into two separate styrofoam cups. Record $V_C$ and $V_H$.
- Set up an experiment like we did in class...
- In logger pro, start your experiment for 400 seconds, measuring 1 sample per second
- Place a temperature sensor in each cup and wait while the thermometers settle.
- Pour the cold into the hot this time and put both sensors in the cup with the mixture.
- Wait until the sensors equilibrate with the mixture
- Stop Logger Pro
- Examine your graph and your data, and report $T_C$, $T_H$, and $T_f$ of the mixture.
- Then use Eq. $\ref{Tf}$ to calculate the expected final temperature of your mixture.
- Compare your measured $T_f$ and your expected $T_f$. Were they close? Any ideas about why they might have differed a little bit?
Lab 6 - the energy connection
In Lab 6 you found out about the equation which connects the heat energy $Q$ energy absorbed when you change the temperature by
by $\Delta T$ (in degrees C) of something (like water) with a mass, $m$ in grams, which has a heat capacity of $c$ (in calories/g/${}^\circ$C).
$$Q=mc\Delta T.$$
This gives us another way to think about mixing two quantities of water. Visually let's think of mixing like this:
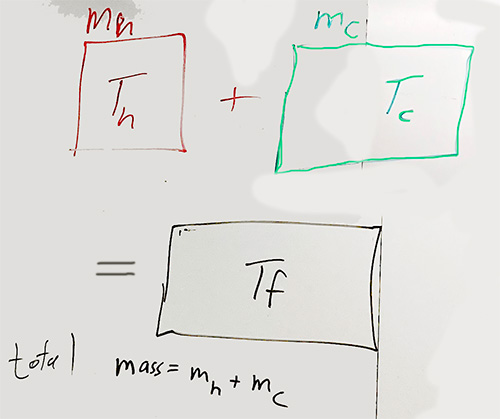
That is, We're going to mix
$m_h$ grams of hot water at an initial temperature $T_h$ and
$m_c$ grams of cold water at an initial temperature $T_c$.
After they're mixed together, the final mixture has a total mass of $m_h+mc$ and the final temperature is somewhere between $T_c$ and $T_f$, that is $T_c \lt T_f \lt T_h$.
The hot water started at $T_h$, but in the process of mixing it ended up at a lower temperature $T_f$. That is to say, it cooled down, or its temperature changed by an amound $(T_h-T_f)$ as a result of coming in contact with the cold water. The hot water lost an amount of thermal energy: $$Q_\text{lost by hot}=m_hc(T_h-T_f)$$
Meanwhile, the cold water started at $T_c$, but in the process of mixing it ended up at a higher temperature $T_f$. That is to say, it heated up, or its temperature changed by an amound $(T_f-Tc)$ as a result of coming in contact with the hot water. The cold water gained an amount of thermal energy: $$Q_\text{gained by cold}=m_c(T_f-T_c)$$ Where did the energy come from to heat the cold water? It could have gained the energy that the hot water lost! Let's imagine that just as much energy was lost by the hot water as the amount gained by the cold water. This means we set the two heats equal: $$\begineq Q_\text{gained by cold} &= Q_\text{lost by hot}\\ m_cc(T_f-T_c)&=m_hc(T_h-T_f)\\ m_c(T_f-T_c)&=m_h(T_h-T_f)\\ \endeq$$ $c$ is the heat capacity of water, and it's the same number, 1 calorie / gram / ${}^\circ$C, whether the water is hot or cold. So in the last step I've used some algebra: I've divided both the left and the right side of the equation by $c$ to cancel out the identical $c$ on either side.
Now, in the lab 5 you measured all the masses and temperatures of the hot and cold water. So it seems like we could try to solve this equation for $t_f$ to see what this business of setting the energies equal predicts for the final temperature. Maybe / Hopefully it will give us the same thing as what we found above in equation ($\ref{Tf}$)!?
So, prepare to do algebra!: The basic idea is whatever you do to the left side of an equation you should also do to the right side. I'll guide you through the steps we'll take and show you what's happening on the left, and you should do the same sort of thing on the right side. We'll tryto get only things with $T_f$ in them on the left, and every thing else on the right.
- First, let's carry out the multiplication on the left and the right: $$\begineq m_c(T_f-T_c)&=m_h(T_h-T_f)\\ m_cT_f -m_cT_c&=m_hT_h-m_hT_f\\ \endeq$$
- Let's move the factor with $T_c$ in it to the right side, by adding $+m_cT_c$ to both sides... like this... $$\begineq m_cT_f -m_cT_c&=(m_hT_h-m_hT_f)\\ m_cT_f -m_cT_c\color{red}+m_cT_c&=(...)\color{red}+m_cT_c\\ m_cT_f &= \ ?+?+? \\ \endeq$$ The sum of the two factors with $T_c$ in them was zero. So we're left with just one factor, with a $T_f$ in it on the left, and three factors on the right.
- I hope that one of your three factors on the right has a $T_f$ in it! We want to bring that one to the left side, and we can do it by adding its opposite. This should be accomplished by... $$\begineq m_cT_f \color{red}+m_hT_f&= \ ?+?+? \ ]\+\color{red}+m_hT_f \\ m_cT_f +m_hT_f&= \ ?+? \\ \endeq$$ Now you should have cancelled out one of the three factors on the right leaving just two factors, with no $T_f$ in any thing.
- Now, I notice that both factors on the left have a $T_f$ in them so I'll just re-arrange things on the left, without doing anything to the right: $$ \begineq m_cT_f +m_hT_f&= \ ?+? \\ (m_c +m_h)T_f&= \ ?+? \\ \endeq$$
- Ah, we're ready to completely isolate $T_f$ on the left! I can divide both sides by $(m_c+m_h)$: $$ \begineq (m_c +m_h)T_f&= \ ?+? \\ \frac{(m_c +m_h)}{\color{red}(m_c +m_h)}T_f&= \frac{?+?}{\color{red}(m_c +m_h)} \\ T_f&= \frac{?+?}{(m_c +m_h)} \\ T_f&= \frac{?}{(m_c +m_h)} +\frac{?}{(m_c +m_h)} \\ \endeq$$
Compare the expression you ended up with to Eq ($\ref{Tf}$)! Remember that a volume of 1 milliliter of water has a mass of 1 gram. Sothe number of ml of water is alway the same as the number of grams that it weighs. So, you could write all the masses as volumes, or vice versa...
- $m_c\leftrightarrow V_C$,
- $m_h\leftrightarrow V_H$,
Write up your steps to get from our original expression--setting the heat lost by the hot water equal to the heat gained by the cold water-- up to your final expression for $T_f$ and include that in your writeup for Lab 6.